

|
MONITORING OF PEDIATRIC HIV INFECTION
Immunologic Parameters in Children
Specific Issues in Antiretroviral Therapy for HIV-Infected Adolescents
Specific Issues of Adherence for HIV-Infected Children and Adolescents
Immunologic Parameters in Children
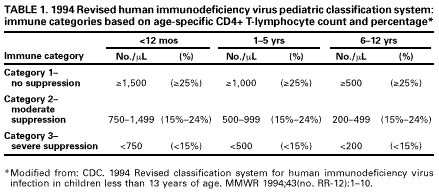
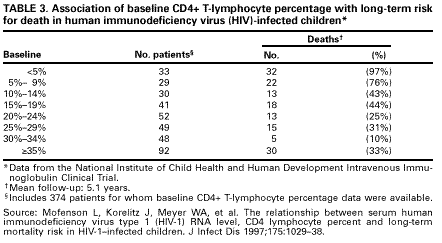
Clinicians interpreting CD4+ T-lymphocyte number for children must consider age as a variable. CD4+ T-lymphocyte count and percentage values in healthy infants who are not infected with HIV are considerably higher than those observed in uninfected adults and slowly decline to adult values by age 6 years (22,23). A pediatric clinical and immunologic staging system for HIV infection has been developed that includes agerelated definitions of immune suppression (Tables 1 and 2) (24). Although the CD4+ absolute number that identifies a specific level of immune suppression changes with age, the CD4+ percentage that defines each immunologic category does not. Thus, a change in CD4+ percentage, not number, may be a better marker of identifying disease progression in children. In infected children and adults, the CD4+ cell count declines as HIV infection progresses, and patients with lower CD4+ cell counts have a poorer prognosis than patients with higher counts (Table 3).
Because knowledge of immune status (i.e., CD4+ T-lymphocyte count and percentage) is essential when caring for HIV-infected infants and children, CD4+ T-lymphocyte values should be obtained as soon as possible after a child has a positive virologic test for HIV and every 3 months thereafter (25,26). Infected infants who have a thymic defect lymphocyte immunophenotypic profile (i.e., CD4+ count <1,900/mm3 and CD8+ count >850/mm 3) during the first 6 months of life have had more rapid HIV disease progression than infants who do not have this profile (27).
The CD4+ T-lymphocyte count or percentage value is used in conjunction with other measurements to guide antiretroviral treatment decisions and primary prophylaxis for PCP after age 1 year. However, measurement of CD4+ cell values can be associated with considerable intrapatient variation. Even mild intercurrent illness or the receipt of vaccinations can produce a transient decrease in CD4+ cell number and percentage; thus, CD4+ values are best measured when patients are clinically stable. No modification in therapy should be made in response to a change in CD4+ cell values until the change has been substantiated by at least a second determination, with a minimum of 1 week between measurements.

HIV RNA in Children
Viral burden in peripheral blood can be determined by using quantitative HIV RNA assays. During the period of primary infection in adults, HIV RNA copy number initially rises to high peak levels. Coincident with the body’s humoral and cell-mediated immune response, RNA levels decline by as much as 2-3 log10 copies to reach a stable lower level (i.e., the virologic set point) approximately 6-12 months following acute infection, reflecting the balance between ongoing viral production and immune elimination (28,29). Several studies conducted among adults have indicated that infected persons with lower HIV copy number at the time of RNA stabilization have slower progression and improved survival compared with those with high HIV RNA set points (30, 31). On the basis of such data, recommendations for the use of HIV RNA copy number in deciding to initiate and change antiretroviral therapy in infected adults have been developed (5). These recommendations also are applicable to infected adolescents, particularly those who have acquired HIV infection recently rather than through perinatal infection. These recommendations also are likely to be applicable to perinatally infected children aged >3 years.
The HIV RNA pattern in perinatally infected infants differs from that in infected adults. High HIV RNA copy numbers persist in infected children for prolonged periods (32,33). In one prospective study, HIV RNA levels generally were low at birth (i.e., <10,000 copies/mL), increased to high values by age 2 months (most infants had values >100,000 copies/mL, ranging from undetectable to nearly 10 million copies/mL), and then decreased slowly; the mean HIV RNA level during the first year of life was 185,000 copies/mL (20). Additionally, in contrast to the adult pattern, after the first year of life, HIV RNA copy number slowly declines over the next few years of life (20, 34-36). This pattern probably reflects the lower efficiency of an immature but developing immune system in containing viral replication and possibly a greater number of HIV-susceptible cells.
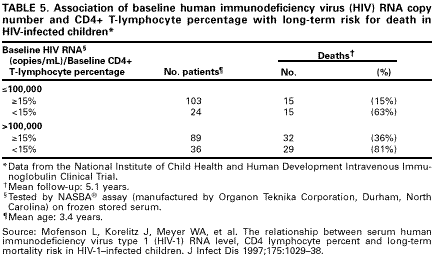
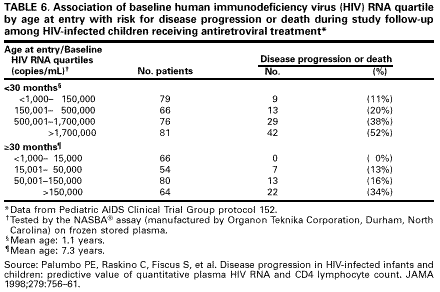
Recent data indicate that high HIV RNA levels (i.e., >299,000 copies/mL) in infants aged <12 months may be correlated with disease progression and death; however, RNA levels in infants who have rapid disease progression and those who do not have overlapped considerably (20, 33). High RNA levels (i.e., levels of >100,000 copies/mL) in infants also have been associated with high risk for disease progression and mortality, particularly if CD4+ T-lymphocyte percentage is <15% (Tables 4 and 5) (35).
Similar findings have been reported in a preliminary analysis of data from PACTG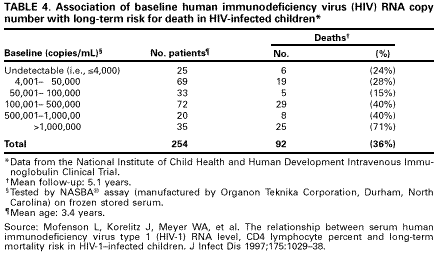
Despite data indicating that high RNA levels are associated with disease progression, the predictive value of specific HIV RNA levels for disease progression and death for an individual child is moderate (35). HIV RNA levels may be difficult to interpret during the first year of life because levels are high and there is marked overlap in levels between children who have and those who do not have rapid disease progression (32). Additional data indicate that CD4+ T-lymphocyte percentage and HIV RNA copy number at baseline and changes in these parameters over time assist in determining the mortality risk in infected children, and the use of the two markers together may more accurately define prognosis (35,36). Similar data and conclusions recently have been reported from several studies involving infected adults (37-39).
Methodologic Considerations in the Interpretation and Comparability of HIV RNA Assays
Most of the published data regarding HIV RNA in children have been obtained using frozen, stored plasma and serum specimens. Some degradation of HIV RNA occurs with specimen storage and delay in specimen processing; thus, the published data on HIV RNA levels in infected children may not be directly comparable with data obtained from specimens that undergo immediate testing (e.g., specimens obtained for patient care). The HIV RNA assays used also differ by study. Therefore, direct extrapolation of the predictive value of HIV RNA levels reported in published studies to HIV RNA assays performed for clinical care purposes may be problematic. Information from ongoing prospective studies will assist in the interpretation of HIV RNA levels among infected infants and children.
The use of HIV RNA assays for clinical purposes requires specific considerations (40), which are discussed more completely elsewhere (5). Several different methods can be used for quantitating HIV RNA, each with different levels of sensitivity; although the results of the assays are correlated, the absolute HIV RNA copy number obtained from a single specimen tested by two different assays can differ by twofold (0.3 log10) or more. For example, plasma RNA measured by the quantitative PCR assay (Amplicor HIV-1 Monitor™, manufactured by Roche Diagnostics Systems, Nutley, New Jersey) yields absolute values approximately twice (0.3 log10) those obtained using a signal amplification, branched-chain DNA assay (Quantiplex ® , manufactured by Chiron Corporation, Emeryville, California) (5,41,42). Similarly, plasma RNA measured by the nucleic acid sequence-based amplification assay (NASBA ® , manufactured by Organon Technika, Durham, North Carolina) yields absolute values approximately twice those obtained using the Quantiplex ® assay but values relatively comparable with those obtained using the Amplicor HIV-1 Monitor ‘ assay (41-43). Therefore, one HIV RNA assay method should be used consistently for monitoring each patient. Choice of HIV RNA assay, particularly for young children, may be influenced by the amount of blood required for the assay. The NASBA ® assay requires the least amount of blood (i.e., 100 mL of plasma), followed by the Amplicor HIV-1 Monitor ‘ (i.e., 200 mL of plasma) and the Quantiplex ® assays (i.e., 1 mL of plasma).
Biologic variation in HIV RNA levels within one person is well documented, and repeated measurement of HIV RNA levels in a clinically stable infected adult can vary by as much as threefold (0.5 log10) in either direction over the course of a day or on different days (5,39,44). This biologic variation may be greater in infected infants and young children. In children with perinatally acquired HIV infection, RNA copy number slowly declines even without therapy during the first several years after birth, although it persists at higher levels than those observed in most infected adults (20, 34, 35). This decline is most rapid during the first 12-24 months after birth, with an average decline of approximately 0.6 log10 per year; a slower decline continues until approximately age 4-5 years (average decline of 0.3 log10 per year). This inherent biologic variability must be considered when interpreting changes in RNA copy number in children. Thus, only changes greater than fivefold (0.7 log10) in infants aged <2 years and greater than threefold (0.5 log10) in children aged >2 years after repeated testing should be considered reflective of a biologically and clinically substantial change. To reduce the impact of assay variability in the clinical management of patients, two samples can be obtained at baseline and the average of the two values used for comparison with future tests. No alteration in therapy should be made as a result of a change in HIV copy number unless the change is confirmed by a second measurement. Because of the complexities of HIV RNA testing and the agerelated changes in HIV RNA in children, interpretation of HIV RNA levels for clinical decision making should be done by or in consultation with an expert in pediatric HIV infection.
Specific Issues in Antiretroviral Therapy for HIV-Infected Adolescents Adult guidelines for antiretroviral therapy are appropriate for post-pubertal adolescents because HIV-infected adolescents who were infected sexually or through injecting drug use during adolescence follow a clinical course that is more similar to that of adults than to that of children (5). The immunopathogenesis and virologic course of HIV infection in adolescents is being defined. Most adolescents have been infected during their teenage years and are in an early stage of infection, making them ideal candidates for early intervention. A limited but increasing number of HIV-infected adolescents are long-term survivors of HIV infection acquired perinatally or through blood products as young children. Such adolescents may have a unique clinical course that differs from that of adolescents infected later in life (45). Because many adolescents with HIV infection are sexually active, issues associated with contraception and prevention of HIV transmission should be discussed between the healthcare provider and the adolescent.
Dosage for medications for HIV infection and opportunistic infections should be prescribed according to Tanner staging of puberty (46) and not on the basis of age (25). Adolescents in early puberty (i.e., Tanner Stage I and II) should be administered doses using pediatric schedules, whereas those in late puberty (i.e., Tanner Stage V) should follow adult dosing schedules. Youth who are in their growth spurt (i.e., in females, Tanner Stage III and in males, Tanner Stage IV) should be closely monitored for medication efficacy and toxicity when using adult or pediatric dosing guidelines. Puberty is a time of somatic growth and sex differentiation, with females developing more body fat and males more muscle mass. Although these physiologic changes theoretically could affect drug pharmacokinetics (especially for drugs with a narrow therapeutic index that are used in combination with protein bound medicines or hepatic enzyme inducers or inhibitors), no clinically consequential impact has been noted with nucleoside analogue reverse transcriptase inhibitor (NRTI) antiretroviral drugs (47). Clinical experience with protease inhibitors and non-nucleoside reverse transcriptase inhibitor antiretroviral drugs is more limited.
Specific Issues of Adherence for HIV-Infected Children and Adolescents
Lack of adherence to prescribed regimens and subtherapeutic levels of antiretroviral medications may enhance the development of drug resistance. Data indicate that the development of resistance to one of the available protease inhibitor antiretrovirals may reduce susceptibility to some or all of the other available protease inhibitor drugs, thus substantially reducing subsequent treatment options. Therefore, education of infected children and/or their caregivers regarding the importance of compliance with the prescribed drug regimen is necessary when therapy is initiated and should be reinforced during subsequent visits. Many strategies can be used to increase medication adherence, including intensive patient education over several visits before therapy is initiated, the use of cues and reminders for administering drugs, development of patient focused treatment plans to accommodate specific patient needs, and mobilization of social and community support services.
Adherence to drug regimens is especially problematic for children. Infants and young children are dependent on others for administration of medication; thus, assessment of the capacity for adherence to a complex multidrug regimen requires evaluation of the caregivers and their environments and the ability and willingness of the child to take the drug. Liquid formulations or formulations suitable for mixing with formula or food are necessary for administration of oral drugs to young children. Lack of palatability of such formulations can be problematic depending on the child’s willingness and ability to accept and retain the medication. Absorption of some antiretroviral drugs can be affected by food, and attempting to time the administration of drugs around meals can be difficult for caregivers of young infants who require frequent feedings. Many other barriers to adherence to drug regimens exist for children and adolescents with HIV infection. For example, unwillingness of the caregivers to disclose their child’s HIV infection status to others may create specific problems, including reluctance of caregivers to fill prescriptions in their home neighborhood, hiding or relabeling medications to maintain secrecy within the home, reduction of social support (a variable associated with diminished treatment adherence), and a tendency to eliminate midday doses when the parent is away from the home or the child is at school.
A comprehensive assessment of adherence issues should be instituted for all children in whom antiretroviral treatment is considered; evaluations should include nursing, social, and behavioral assessments. Intensive follow up is required particularly during the critical first few months after therapy is started; patients should be seen frequently to assess adherence, drug tolerance, and virologic response. Coordinated, comprehensive, family-centered systems of care often can address many of the daily problems facing families that may affect adherence to complex medical regimens. For some families, certain issues (e.g., a safe physical environment and adequate food and housing) may take priority over medication administration and need to be resolved. Case managers, mental health counselors, peer educators, outreach workers, and other members of the multidisciplinary team often may be able to address specific barriers to adherence.
HIV-infected adolescents have specific adherence problems. Comprehensive systems of care are required to serve both the medical and psychosocial needs of HIV-infected adolescents, who are frequently inexperienced with healthcare systems. Many HIV-infected adolescents face challenges in adhering to medical regimens for reasons that include a) denial and fear of their HIV infection; b) misinformation; c) distrust of the medical establishment; d) fear and lack of belief in the effectiveness of medications; e) low self esteem; f) unstructured and chaotic lifestyles; and g) lack of familial and social support. Treatment regimens for adolescents must balance the goal of prescribing a maximally potent antiretroviral regimen with realistic assessment of existing and potential support systems to facilitate adherence.
Developmental issues make caring for adolescents unique. The adolescent’s approach to illness is often different from that of adults. The concrete thought processes of adolescents make it difficult for them to take medications when they are asymptomatic, particularly if the medications have side effects. Adherence with complex regimens is particularly challenging at a time of life when adolescents do not want to be different from their peers. Further difficulties face adolescents who live with parents to whom they have not yet disclosed their HIV status and those who are homeless and have no place to store medicine.
Go to the NIH Guidelines for Pediatric Antiretroviral Use Menu
Go to the NIH Report & Guidelines Menu
Go to the HIVpositive.us Main Menu