
|
NIH Guidelines of Therapy
Guidelines for the Use of Antiretroviral Agents in HIV-Infected Adults
and Adolescents
12. APPENDICES


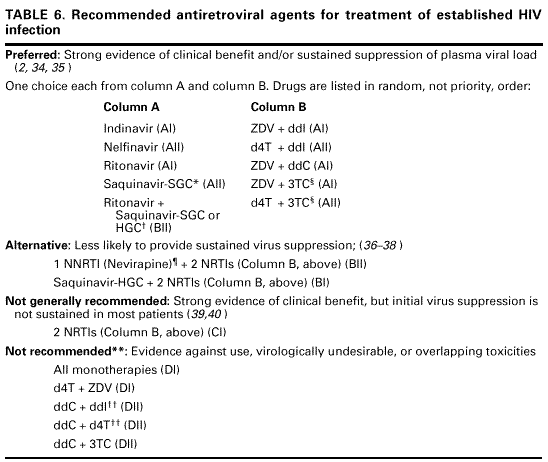
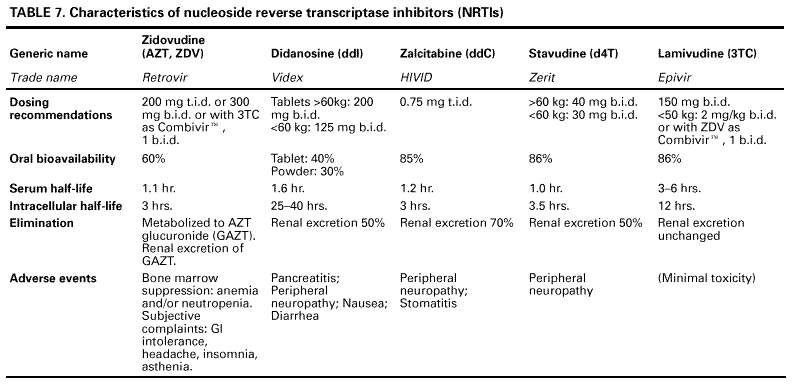
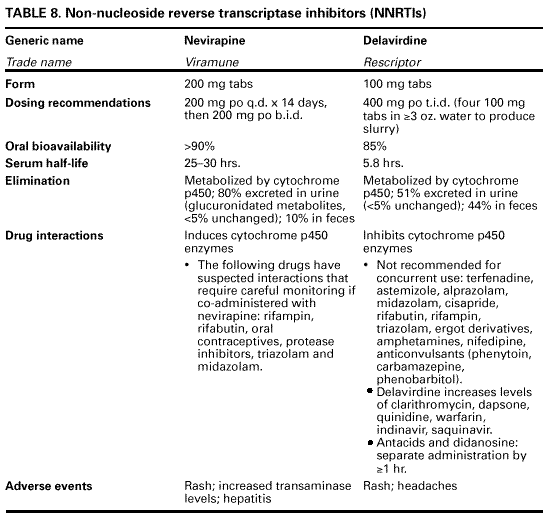
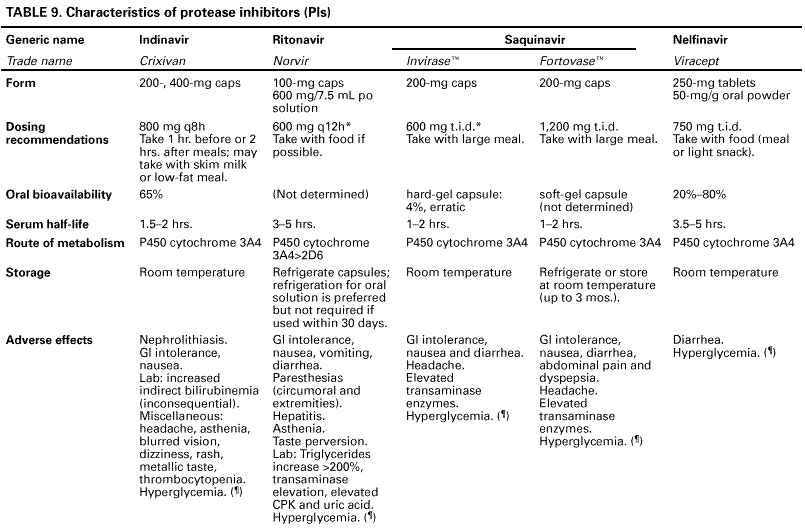
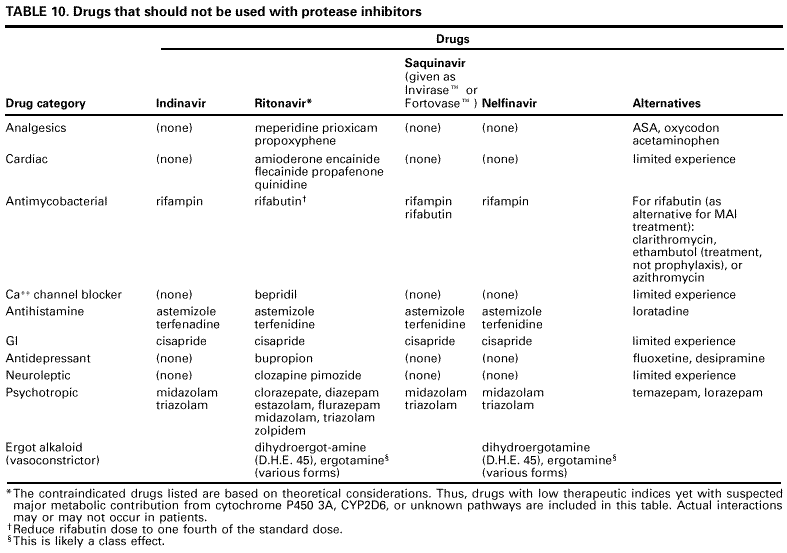
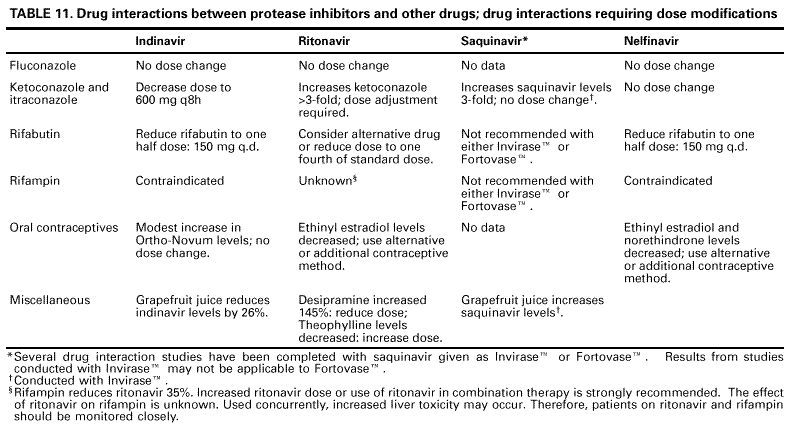
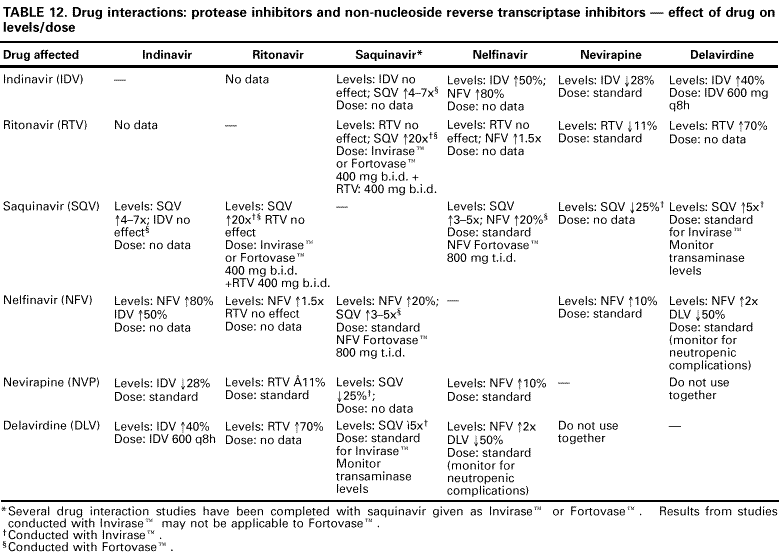
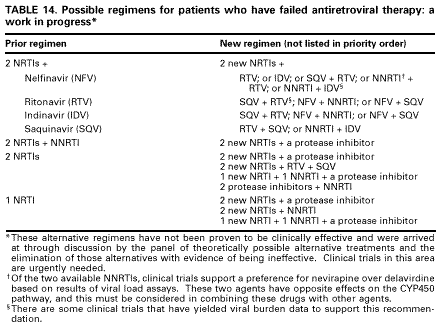
Table 13. Guidelines for changing an antiretroviral regimen
for suspected drug failure
- Criteria for changing therapy include a suboptimal reduction
in plasma viremia after initiation of therapy, reappearance of viremia
after suppression to undetectable, substantial increases in plasma viremia
from the nadir of suppression, and declining CD4 + T cell numbers. Refer
to the more extensive discussion of these criteria in “Criteria for Changing
Therapy” on pages 53 & 54.
- When the decision to change therapy is based on viral load
determination, it is preferable to confirm with a second viral load test.
- Distinguish between the need to change a regimen because
of drug intolerance or inability to comply with the regimen versus failure
to achieve the goal of sustained viral suppression; single agents can be
changed or dose reduced in the event of drug intolerance.
- In general, do not change a single drug or add a single
drug to a failing regimen; it is important to use at least two new drugs
and preferably to use an entirely new regimen with at least three new drugs.
- Many patients have limited options for new regimens of
desired potency; in some of these cases, it is rational to continue the
prior regimen if partial viral suppression was achieved.
- In some cases, regimens identified as suboptimal for initial
therapy are rational due to limitations imposed by toxicity, intolerance,
or nonadherence. This especially applies in late-stage disease. For patients
with no rational alternative options who have virologic failure with return
of viral load to baseline (pretreatment levels) and a declining CD4+T cell
count, discontinuation of antiretroviral therapy should be considered.
- Experience is limited with regimens using combinations
of two protease inhibitors or combinations of protease inhibitors with
nevirapine or delavirdine; for patients with limited options due to drug
intolerance or suspected resistance, these regimens provide possible alternative
treatment options.
- There is limited information about the value of restarting
a drug that the patient has previously received. The experience with zidovudine
is that resistant strains are often replaced with “wild-type” zidovudine
sensitive strains when zidovudine treatment is stopped, but resistance
recurs rapidly if zidovudine is restarted. Although preliminary evidence
indicates that this occurs with indinavir, it is not known if similar problems
apply to other nucleoside analogues, protease inhibitors, or NNRTIs, but
a conservative stance is that they probably do.
- Avoid changing from ritonavir to indinavir or vice versa
for drug failure, because high-level cross-resistance is likely.
-
Avoid changing from nevirapine to delavirdine or vice versa for drug failure,
because high-level cross-resistance is likely.
- The decision to change therapy and the choice of a new
regimen require that the clinician have considerable expertise in the care
of persons living with HIV infection. Physicians who are less experienced
in the care of persons with HIV infection are strongly encouraged to obtain
assistance through consultation with or referral to a clinician who has
considerable expertise in the care of HIV-infected patients.

Return to the top of this page
Go to the NIH Report & Guidelines Menu
Go to the NIH Guidelines Menu
Go to the NIH Report Menu
Go to the Treatment Menu
Go to the HIVpositive.us Main Menu

















